Abstract
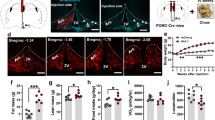
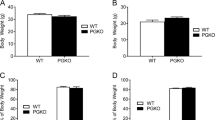
In response to moderately increased dietary fat content, melanocortin-4 receptor-null mutant (MC4R−/−) mice exhibit hyperphagia and accelerated weight gain compared to wild-type mice. An increased feed efficiency (weight gain/kcal consumed) argues that mechanisms in addition to hyperphagia are instrumental in causing weight gain. We report two specific defects in coordinating energy expenditure with food intake in MC4R−/− mice. Wild-type mice respond to an increase in the fat content of the diet by rapidly increasing diet-induced thermogenesis and by increasing physical activity, neither of which are observed in MC4R−/− mice. Leptin-deficient and MC3R−/− mice regulate metabolic rate similarly to wild-type mice in this protocol. Melanocortinergic pathways involving MC4-R-regulated neurons, which rapidly respond to signals not requiring changes in leptin, thus seem to be important in regulating metabolic and behavioral responses to dietary fat.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Leibel, R. L., Chung, W. K. & Chua, S. C .J. The molecular genetics of rodent single gene obesities. J. Biol. Chem. 272, 31937–31940 (1997).
Yen, T. T., McKee, M. M. & Stamm, N. B. Thermogenesis and weight control. Int. J. Obes. 8 (Suppl. 1), 65–78 (1984).
Frigeri, L. G., Wolff, G. L. & Teguh, C. Differential responses of yellow Avy/A and agouti A/a (BALB/c X VY) F1 hybrid mice to the same diets: glucose tolerance, weight gain, and adipocyte cellularity. Int. J. Obes. 12, 305–320 (1988).
Marie, L. S., Miura, G. I., Marsh, D. J., Yagaloff, K. & Palmiter, R. D. A metabolic defect promotes obesity in mice lacking melanocortin-4 receptors. Proc. Natl. Acad. Sci. USA 97, 12339–12344 (2000).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Cowley, M. A. et al. Integration of NPY, AGRP, and melanocortin signals in the hypothalamic paraventricular nucleus: evidence of a cellular basis for the adipostat. Neuron 24, 155–163 (1999).
Marsh, D. J. et al. Response of melanocortin-4 receptor-deficient mice to anorectic and orexigenic peptides. Nat. Genet. 21, 119–122 (1999).
Halaas, J. L. et al. Physiological response to long-term peripheral and central leptin infusion in lean and obese mice. Proc. Natl. Acad. Sci. USA 94, 8878–8883 (1997).
Boston, B. A., Blaydon, K. M., Varnerin, J. & Cone, R. D. Independent and additive effects of central POMC and leptin pathways on murine obesity. Science 278, 1641–1644 (1997).
Cheung, C. C., Clifton, D. K. & Steiner, R. A. Proopiomelanocortin neurons are direct targets for leptin in the hypothalamus. Endocrinology 138, 4489–4492 (1997).
Seeley, R. J. et al. Melanocortin receptors in leptin effects. Nature 390, 349 (1997).
Satoh, N. et al. Satiety effect and sympathetic activation of leptin are mediated by hypothalamic melanocortin system. Neurosci. Lett. 249, 107–110 (1998).
Smith, A. I. & Funder, J. W. Proopiomelanocortin processing in the pituitary, central nervous system, and peripheral tissues. Endocr. Rev. 9, 159–179 (1988).
Low, M. J., Simerly, R. D. & Cone, R. D. Receptors for the melanocortin peptides in the central nervous system. Curr. Opin. Endocrinol. Diabet. 1, 79–88 (1994).
Baskin, D. G. et al. Insulin and leptin: dual adiposity signals to the brain for the regulation of food intake and body weight. Brain. Res. 848, 114–123 (1999).
Hagan, M. M. et al. Role of the CNS melanocortin system in the response to overfeeding. J. Neurosci. 19, 2362–2367 (1999).
Enerback, S. et al. Mice lacking mitochondrial uncoupling protein are cold-sensitive but not obese. Nature 387, 90–94 (1997).
Butler, A. A. et al. A unique metabolic syndrome causes obesity in the melanocortin-3 receptor-deficient mouse. Endocrinology 141, 3518–3521 (2000).
Schutz, Y., Bessard, T. & Jecquier, E. Thermogenesis measured over a whole day in obese and non-obese women. Am. J. Clin. Nutr. 40, 542–552 (1984).
Ferrannini, E. The theoretical basis of indirect calorimetry: a review. Metabolism 37, 287–301 (1988).
Zurlo, F. et al. Low ratio of fat to carbohydrate oxidation as predictor of weight gain: study of 24-h RQ. Am. J. Phys. 259, E650–657 (1990).
Nakaya, Y. et al. Respiratory quotient in patients with non-insulin-dependent diabetes mellitus treated with insulin and oral hypoglycemic agents. Ann. Nutr. Metab. 42, 333–340 (1998).
Mercer, S. W. & Trayhurn, P. Effect of high fat diets on energy balance and thermogenesis in brown adipose tissue of lean and genetically obese ob/ob mice. J. Nutr. 117, 2147–2153 (1987).
Himms-Hagen, J., Hogan, S. & Zaror-Behrens, G. Increased brown adipose tissue thermogenesis in obese (ob/ob) mice fed a palatable diet. Am. J. Physiol. 250, E274–E281 (1986).
Collins, S. et al. Role of leptin in fat regulation. Nature 380, 677 (1996).
Levine, J. A., Eberhardt, N. L. & Jensen, M. D. Role of nonexercise activity thermogenesis in resistance to fat gain in humans. Science 283, 212–214 (1999).
Trayhurn, P., Jones, P. M., McGuckin, M. M. & Goodbody, A. E. Effect of overfeeding on energy balance and brown fat thermogenesis in obese (ob/ob) mice. Nature 295, 323–325 (1982).
Surwit, R. S., Kuhn, C. M., Cochrane, C., McCubbin, J. A. & Feinglos, M. N. Diet-induced type II diabetes in C57Bl/6J mice. Diabetes 37, 1163–1167 (1988).
Saladin, R. et al. Transient increase in obese gene expression after food intake or insulin administration. Nature 377, 527–532 (1995).
Haynes, W. G., Morgan, D. A., Djalali, A., Sivitz, W. I. & Mark, A.L. Interactions between the melanocortin system and leptin in control of sympathetic nerve traffic. Hypertension 33, 542–547 (1999).
De Souza, J., Butler, A. A. & Cone, R. D. Disproportionate inhibition of feeding in A(y) mice by certain stressors: a cautionary note. Neuroendocrinology 72, 126–132 (2000).
Ziotopoulou, M., Mantzoros, C. S., Hileman, S. M. & Flier, J. S. Differential expression of hypothalamic neuropeptides in the early phase of diet-induced obesity in mice. Am. J. Physiol. Endocrinol. Metab. 279, E838–E845 (2000).
Eckel, L. A. et al. Chronic administration of OB protein decreases food intake by selectively reducing meal size in female rats. Am. J. Physiol. 275, R186–193 (1998).
Loftus, T. M. et al. Reduced food intake and body weight in mice treated with fatty acid synthase inhibitors. Science 288, 2379–2381 (2000).
Acknowledgements
This work was supported by NIH grant PPGDK55819 to R.D.C. We thank K. Khong, E. Douthit and K.E. Miles for their help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Butler, A., Marks, D., Fan, W. et al. Melanocortin-4 receptor is required for acute homeostatic responses to increased dietary fat. Nat Neurosci 4, 605–611 (2001). https://doi.org/10.1038/88423
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/88423
This article is cited by
-
A hypothalamomedullary network for physiological responses to environmental stresses
Nature Reviews Neuroscience (2022)
-
Pathophysiological mechanisms leading to muscle loss in chronic kidney disease
Nature Reviews Nephrology (2022)
-
Dietary switch to Western diet induces hypothalamic adaptation associated with gut microbiota dysbiosis in rats
International Journal of Obesity (2021)
-
Oxytocin Neurons Enable Melanocortin Regulation of Male Sexual Function in Mice
Molecular Neurobiology (2019)
-
Central regulation of brown adipose tissue thermogenesis and energy homeostasis dependent on food availability
Pflügers Archiv - European Journal of Physiology (2018)