Abstract
The best-known situation indissolubly linked to mosaicism is the uniparental disomy where a trisomic or monosomic zygote develops at least one cell line with 46 chromosomes. The mosaicism normal/abnormal cell lines may remain confined to placenta or persist in the embryo. Here, we describe a second situation that might also be indissolubly linked to a mosaic condition or at least to a confined placental mosaicism. We analysed the case of a mosaicism del(8p)/inv dup(8p) found in prenatal diagnosis. We had already demonstrated that the first product of the abnormal meiotic recombination at the basis of the inv dup rearrangements is a dicentric chromosome. Its breakage leads to the formation of a deleted and an inv dup chromosome. Although we had previously assumed that the dicentric underwent a breakage at meiosis II so that the zygote inherited the inv dup chromosome, our findings and those of others indeed indicate that the dicentric may be inherited in the zygote and that it might persist as such in early postzygotic stages, then undergoing different breakages in different cells leading to different abnormal chromosomes, either deleted or duplicated. Selection versus the most viable cell line(s) results either in a confined placental mosaicism with the inv dup cell line as the only one present in the embryo or in children with both the deleted and the inv dup cell lines. Phenotype/karyotype relationships in inv dup rearrangements must also take into account the influence of the other abnormal cell line during embryogenesis.
Similar content being viewed by others
Introduction
The molecular characterization of several rearrangements interpreted as simple duplications led to the discovery that most of them were in fact inverted duplications associated with the deletion of the portion distal to the duplication. The best-known case is that of the inv dup(8p).1, 2 Other examples are those concerning 1q,3, 4 2q,5 3p6, 7 4p,8 5p,9, 10 9p,11 10p and 10q,12 18p13 18q,14 21q15 and the X;X or the Y;Y rearrangements leading to duplications of parts of the short or the long arm with concomitant deletion.16 It has been assumed1, 5 that the first product of the abnormal meiotic recombination on the basis of this type of rearrangement was a dicentric chromosome, either p → q∷q → p or q → p∷p → q. The dicentric, due to its intrinsic instability, would undergo an asymmetric breakage between the two centromeres leading to the final inv dup. In the case of the inv dup(8p), the breakage of the original dicentric chromosome 8qter → 8p23.1∷8p23.1 → 8qter occurs in different positions between the two centromeres with a preferential breakpoint at the level of the second centromere that became inactive (six out of 16 cases).1 In no other case of autosomal inv dups two alphoid signals have been reported; this seems to be due to the fact that very large duplications are unviable or that the second centromere has not been detected due to the use of staining techniques inappropriate in highlighting the centromere. Previously, we had assumed1, 5 that the dicentric's breakage occurred at the second meiotic division leading to a gamete having the inv dup(8p) and a gamete deleted for a portion of 8p. However, two recent papers17, 18 and an older one19 made us re-evaluate the entire situation suggesting that the breakage of the dicentric might not occur at meiosis II but that, at least in some cases, the dicentric could be inherited as such in the zygote, after which it undergoes to breakage, thus, leading to a mosaic situation of the conception's product. The mosaicism consists in a cell line containing the inv dup(8p) and a second cell line containing either the deleted 8p or other products derived from the deletion of the dicentric. The finding of a new mosaic inv dup(8p) case indeed made us hypothesize that mosaicism or at least confined placental mosaicism might be the rule for all inv dup rearrangements.
Case report
A 32-year-old healthy woman was referred between the 11th and 12th week of pregnancy for cytogenetic evaluation after abnormal fetal ultrasound findings showing subcutaneous oedema in the neck region with generalized fetal hydrops associated with heart malformation. Transabdominal and transvaginal echocardiography with colour doppler flow imaging showed atrial and ventricular septal defect, dilated left ventricle and pericardial effusion. The couple decided for the termination of pregnancy at the end of the 12th week, after the finding of the 8p deletion on the direct chorionic villus sample.
Materials and methods
Cytogenetic analysis was performed on a 12-week chorionic villus sample (CVS) both after 24 h incubation (direct CVS) and after 11 days of culturing (cultured CVS). Other metaphases were analysed on slides obtained from other long-term cultures. FISH with subtelomeric probes was carried out following the manufacturer's (Vysis) instructions. Single- and dual-colour FISH analysis was performed as described.2 BAC DNA extraction was carried out using the PhasePrep BAC DNA kit (SIGMA). BAC clones were labelled with biotin-16dUTP and digoxigenin-dUTP by nick translation (BRL Life Technologies). Signals from biotin-labelled probes were developed using alternate layers of avidin–fluorescein isothiocyanate (avidin–FITC) and biotinylated anti-avidin. Signals from digoxin-labelled probes were developed using alternate layers of mouse-antidigoxigenin, avidin-alpha-mouse and antiavidin-alpha-rabbit antibodies. Slides were counterstained with DAPI (Sigma) and mounted in Vectashield antifading medium (Vector Laboratories, USA). Signals were visualized under a Nikon E1000 microscope equipped with a cooled charge-coupled device (CCD) camera and Genikon image analysis software. Genotyping of 8p polymorphic loci from the DNAs of the CVS and the parents was performed by amplification with primers (Table 1) labelled with fluorescent probes (ABI 5-Fam, Hex) followed by analysis on a ABI 3100 Genetic Analyzer (Applied Biosystems). Amplifications were performed with Taq Gold (Applied Biosystems) using standard protocols. Cytogenetic analysis from peripheral lymphocytes of the parents was carried out.
Results
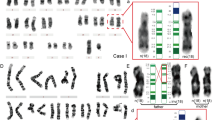
A deletion of the short arm of one chromosome 8 was detected in all the 26 cells that could be analysed from the direct CVS: 46,XX,del(8)(p11. 2) (Figure 1a). Analysis of 20 metaphases obtained from cultured CVS demonstrated the presence of a derivative chromosome 8 with added material on the short arm: 46,XX,der(8)add(8p) (Figure 1b); in three metaphases, the elongated 8p ended with fluorescent satellites (Figure 1c). FISH analysis with probes RP11-13N12 (AC011586), RP11-135I5 (AC022716) (Figure 2a) and chromosome 8 alpha-satellite demonstrated that the der(8) was a monocentric inv dup(8p). A total of 98 metaphases obtained from cultured CVS were counted and in all of them the del(8) was always substituted by the inv dup(8p); no other metaphases with the inv dup(8p) ending with fluorescent satellites were found. To exclude the presence of subtelomere probes specific to other chromosomes at the tip of the inv dup(8p), FISH was carried out demonstrating that no subtelomeric probes were translocated at the tip of the inv dup(8p). Polymorphic markers confirmed that the der(8) was an inv dup(8p) proving the presence of an 8pter deletion and that of duplicated alleles at 8p21–p22. This analysis also showed the maternal origin of the rearrangement (Figure 3). The final interpretation of the cytogenetic analysis on direct CVS and cultured CVS was: 46,XX,del(8)(p11.2)[26]/46,XX,inv dup(8p)[98] with three of the inv dup(8p) metaphases having a satellite translocated to the short arm of the inv dup(8p). Both parents had a normal karyotype on 550 bands. FISH with probes RP11-399J23 (AC068353) and RP11-589N15 (AC025857) at 3.5 Mb between each other, performed on prometaphases and metaphases from the mother, revealed the presence of the cryptic heterozygous inversion between REPP and REPD (Figure 2b).
(a) Q-banded metaphase from direct CVS showing the del(8)(p11) (arrow) and the normal chromosome (arrowhead). The six small arrows indicate the D-group chromosomes. (b) Q-banded metaphase from cultured CVS showing the inv dup(8p) (arrow) and the normal chromosome (arrowhead). (c): Q-banded metaphase from cultured CVS showing the inv dup(8p) (arrow) with satellites at the tip of its short arm.
Discussion
We describe the case of a 12-week fetus with severe echographic anomalies showing an 8p deletion in the direct CVS and an inv dup(8p) in the cultured CVS. We could not exclude that the inv dup(8p) cell line was present in the direct CVS, since only 26 metaphases could be examined. On the contrary, in the cultured CVS, the absence of the del(8p) cell line was demonstrated through the analysis of 98 metaphases. The rearrangement originated at the maternal meiosis as demonstrated by the presence of two maternal and one paternal allele at some loci of the duplicated region of the inv dup(8p) (see Table 1) and the mother had the typical heterozygous inversion predisposing to the inv dup(8p) rearrangement.2
The finding of different cell lines with a trisomy in direct CVS and normal cells (either with uniparental disomy or really normal) in cultured CVS is fairly common, possibly reflecting the origin of the mosaicism from the correction of a trisomic conception.20 A similar situation of selecting the most viable cell line might have occurred in our case. In fact, there is no doubt that del(8)(p11.2) fetuses are less viable than the inv dup(8p) ones since no living subjects have ever been reported with such an 8p deletion, whereas several cases of inv dup(8p)s are reported, most if not all of them without life-threatening malformations.16 Although it is impossible to say whether the mosaicism we found is a confined placental mosaicism (CPM) with only inv dup(8p) cells present in the fetus, its cardiac abnormalities might indeed suggest the influence of the 8p deletion cell line on embryo development. Heart defects are rather rare in inv dup(8p) cases, whereas they are quite common in deletion of the terminal end of chromosome 8p typically in the form of atrial ventricular septal defects.21, 22 This type of defect in two large unrelated families provided strong evidence for the involvement of GATA4 at 8p23.1.23 GATA4 is in single copy in the inv dup(8p) chromosomes,2 whereas it should be deleted in the cell line with the del(8p). In fact, the fetus we studied had a cardiac septal defect.
Mosaicisms similar to that described here have been reported three times, twice in two children with dysmorphic features and mental retardation18, 19 and a third one in prenatal diagnosis.17 In the latter case, a mosaic karyotype 46,XX,i(8q) /46,XX,del(8)(p11.2) was found in direct CVS, whereas in the follow-up amniocentesis only metaphases with the inv dup(8p) had been found. The authors suggested that the mosaicism del(8)(p11.2)/inv dup(8p) derives from the postzygotic breakage of a dicentric chromosome 8 formed through unequal recombination of the two pairs of olfactory receptor gene-clusters at 8p23.1. Vermeesh et al18 found the mosaicism del(8)/inv dup(8p) in the lymphocytes of their patient but not in the fibroblasts where only the inv dup(8p) was present. Their molecular analysis demonstrated that the del(8) contained a portion of 8p present also in the inv dup(8p), thus, demonstrating that they were not the one mutual of the other as expected if they were formed by breakage of a dicentric chromosome. Based on this observation, they concluded that the inv dup(8p) originated at meiosis, whereas the 8p- originated by an independent terminal deletion event. We think that the inheritance of a dicentric chromosome to the zygote might indeed explain all the mosaicisms found in the published cases and in our case. We have only to assume that the dicentric chromosome is maintained at least in the cells of the blastula, then undergoing different breakages in different cells leading to inv dup(8p)s with different duplicated regions and to del(8p)s with different deleted regions. Since telomerase activity is present in the early stages of fetal development,24 the broken derivatives of the dicentric chromosome can be healed. Cellular selection favouring the cells with a less severe aneuploidy over those with large duplications or deletions is expected to occur as it is known to occur against trisomic cells.25, 26, 27, 28 The isochromosome 8 in some cells of Soler's case17 and the three cells of our inv dup(8p) having satellites at its tip seem to indicate that the dicentric 8 with both the centromeres still active could have been maintained for a certain duration along embryogenesis. In fact, it seems likely to assume that the isochromosome was formed through the fusion of the two chromatids in a neoformed deleted chromosome 8, which could not be healed by the telomerase any more due to the switch-off of its activity. Similarly, the satellited inv dup(8p) might have been formed later in respect to the nonsatellited inv dup(8p) so that it was stabilized by the telomere capture of the short arm of a D- or a G-chromosome. The same situation was reported in an inv dup(8p) stabilized by telomere capture from 18q.29
The importance of the findings we discussed here relies on the fact that such mosaic situations with two or more cell lines, all with a different abnormality of the same chromosome, might not be rare. Beyond the three cases with the inv dup(8p)/del(8p) mosaicism,17, 18, 19 we were able to find in the literature four 5p−/5p+ cases (10; one studied by us) and several cases with a mosaicism consisting of a cell line with a dicentric isochromosome and another one with a deleted chromosome. Examples are three 18p−/18p+ cases,30, 31, 32 one 18q−/18q+ case,33 two Xq−/Xq+ cases, one of them associated also with a 45,X cell line,34, 35 several cases of dicentric Ypter-q11 that are frequently mosaic for a del(Y)(pter-q11) cell line.16
In conclusion, our findings suggest that (1) mosaicism might be rather common in chromosome rearrangements consisting of inversion duplication associated with distal deletion, that is, in those rearrangements having a dicentric chromosome as the first product of the abnormal meiotic recombination; (2) the timing of the dicentric breakage may be placed in early embryogenesis rather than at the meiosis II and (3) phenotype/karyotype relationship in inv dup rearrangements must also take into account the influence of the other abnormal cell line during embryogenesis.
References
Floridia G, Piantanida M, Minelli A et al: The same molecular mechanism at the maternal meiosis I produces mono- and dicentric 8p duplications. Am J Hum Genet 1996; 58: 785–796.
Giglio S, Broman KW, Matsumoto N et al: Olfactory receptor-gene clusters, genomic-inversion polymorphisms, and common chromosome rearrangements. Am J Hum Genet 2001; 68: 874–883.
Mewar R, Harrison W, Weaver DD, Palmer C, Davee MA, Overhauser J : Molecular cytogenetic determination of a deletion/duplication of 1q that results in a trisomy 18 syndrome-like phenotype. Am J Med Genet 1994; 52: 178–183.
De Brasi D, Rossi E, Giglio S et al: Inv dup del (1)(pter–>q44:q44–>q42:) with the classical phenotype of trisomy 1q42-qter. Am J Med Genet 2001; 104: 127–130.
Bonaglia MC, Giorda R, Poggi G et al: Inverted duplications are recurrent rearrangements always associated with a distal deletion: description of a new case involving 2q. Eur J Hum Genet 2000; 8: 597–603.
Jenderny J, Poetsch M, Hoeltzenbein M, Friedrich U, Jauch A : Detection of a concomitant distal deletion in an inverted duplication of chromosome 3. Is there an overall mechanism for the origin of such duplications/deficiencies? Eur J Hum Genet 1998; 6: 439–444.
Kennedy D, Silver MM, Winsor EJ et al: Inverted duplication of the distal short arm of chromosome 3 associated with lobar holoprosencephaly and lumbosacral meningomyelocele. Am J Med Genet 2000; 91: 167–170.
Cotter PD, Kaffe S, Li L, Gershin IF, Hirschhorn K : Loss of subtelomeric sequence associated with a terminal inversion duplication of the short arm of chromosome 4. Am J Med Genet 2001; 102: 76–80.
Sreekantaiah C, Kronn D, Marinescu RC, Goldin B, Overhauser J : Characterization of a complex chromosomal rearrangement in a patient with a typical catlike cry and no other clinical findings of cri-du-chat syndrome. Am J Med Genet 1999; 86: 264–268.
Perfumo C, Cerruti Mainardi P, Cali A et al: The first three mosaic cri du chat syndrome patients with two rearranged cell lines. J Med Genet 2000; 37: 967–972.
Teebi AS, Gibson L, McGrath J, Meyn MS, Breg WR, Yang-Feng TL : Molecular and cytogenetic characterization of 9p- abnormalities. Am J Med Genet 1993; 46: 288–292.
Hoo JJ, Chao M, Szego K, Rauer M, Echiverri SC, Harris C : Four new cases of inverted terminal duplication: a modified hypothesis of mechanism of origin. Am J Med Genet 1995; 58: 299–304.
Moog U, Engelen JJ, de Die-Smulders CE et al: Partial trisomy of the short arm of chromosome 18 due to inversion duplication and direct duplication. Clin Genet 1994; 46: 423–429.
Courtens W, Grossman D, Van Roy N et al: Noonan-like phenotype in monozygotic twins with a duplication-deficiency of the long arm of chromosome 18 resulting from a maternal paracentric inversion. Hum Genet 1998; 103: 497–505.
Pangalos C, Theophile D, Sinet PM et al: No significant effect of monosomy for distal 21q22.3 on the Down syndrome phenotype in ‘mirror’ duplications of chromosome 21. Am J Hum Genet 1992; 51: 1240–1250.
Schinzel A : Catalogue of unbalanced chromosome aberrations in man, 2nd edn. Berlin; New York: de Gruyter, 2001.
Soler A, Sanchez A, Carrio A, Badenas C, Mila M, Borrell A : Fetoplacental discrepancy involving structural abnormalities of chromosome 8 detected by prenatal diagnosis. Prenat Diagn 2003; 23: 319–322.
Vermeesch JR, Thoelen R, Salden I, Raes M, Matthijs G, Fryns JP : Mosaicism del (8p)/inv dup(8p) in a dysmorphic female infant: a mosaic formed by a meiotic error at the 8p OR gene and an independent terminal deletion event. J Med Genet 2003; 40: e93.
van Balkom ID, Hagendoorn J, De Pater JM, Hennekam RC : Partial monosomy 8p and partial trisomy 8p with moderate mental retardation. Genet Couns 1992; 3: 83–89.
Wolstenholme J : Confined placental mosaicism for trisomies 2, 3, 7, 8, 9, 16, and 22: their incidence, likely origins, and mechanisms for cell lineage compartmentalization. Prenat Diagn 1996; 16: 511–524.
Devriendt K, Matthijs G, Van Dael R et al: Delineation of the critical deletion region for congenital heart defects, on chromosome 8p23.1. Am J Hum Genet 1999; 64: 1119–1126.
Giglio S, Graw SL, Gimelli G et al: Deletion of a 5-cM region at chromosome 8p23 is associated with a spectrum of congenital heart defects. Circulation 2000; 102: 432–437.
Garg V, Kathiriya IS, Barnes R et al: GATA4 mutations cause human congenital heart defects and reveal an interaction with TBX5. Nature 2003; 424: 443–447.
Wright WE, Piatyszek MA, Rainey WE, Byrd W, Shay JW : Telomerase activity in human germline and embryonic tissues and cells. Dev Genet 1996; 18: 173–179.
Wolstenholme J, Rooney DE, Davison EV : Confined placental mosaicism, IUGR, and adverse pregnancy outcome: a controlled retrospective U.K. collaborative survey. Prenat Diagn 1994; 14: 345–361.
Kalousek DK, Vekemans M : Confined placental mosaicism. J Med Genet 1996; 33: 529–533.
Robinson WP, Barrett IJ, Bernard L et al: Meiotic origin of trisomy in confined placental mosaicism is correlated with presence of fetal uniparental disomy, high levels of trisomy in trophoblast, and increased risk of fetal intrauterine growth restriction. Am J Hum Genet 1997; 60: 917–927.
Robinson WP : Mechanisms leading to uniparental disomy and their clinical consequences. BioEssays 2000; 22: 452–459.
Kostiner DR, Nguyen H, Cox VA, Cotter PD : Stabilization of a terminal inversion duplication of 8p by telomere capture from 18q. Cytogenet Genome Res 2002; 98: 9–12.
Badalian LO, Mutovin GR, Malygina NA, Petrukhin AS : Rare case of mosaicism for chromosome 18, karyotype: 46, XX, del(18) (p11)/46, XX, i(18q). Genetika 1983; 19: 1912–1915.
Sutton SD, Ridler MA : Prenatal detection of monosomy 18p and trisomy 18q mosaicism with unexpected fetal phenotype. J Med Genet 1986; 23: 258–259.
Stephen GS, Couzin DA, Watt JL, Rankin R : Prenatal diagnosis of a case of 46,XY,18p−/46,XY,18p+ mosaicism. Prenat Diagn 1989; 9: 57–60.
Ausems MG, Bhola SL, Post-Blok CA, Hennekam RC, de France HF : 18q− and 18q+ mosaicism in a mentally retarded boy. Am J Med Genet 1994; 53: 296–299.
Morichon-Delvallez N, Couturier J, Bourdrel V : Inherited Xq duplication due to a zygotic translocation t(X;X)(q23;q27). Ann Genet 1988; 31: 117–119.
Calvano S, de Cillis GP, Croce AI, Perla G, Notarangelo A, Zelante L : A complex mosaicism 45,X/46,X,del(Xq)/46,X,idic(Xq) in a patient with secondary amenorrhea. Ann Genet 2002; 45: 137–140.
Acknowledgements
This work was supported by cofin02-and cofin03-MIUR (to OZ), the Italian Telethon Foundation (GP0247Y01 to OZ) and the Cariplo Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pramparo, T., Giglio, S., Gregato, G. et al. Inverted duplications: how many of them are mosaic?. Eur J Hum Genet 12, 713–717 (2004). https://doi.org/10.1038/sj.ejhg.5201240
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejhg.5201240
Keywords
This article is cited by
-
De novo unbalanced translocations have a complex history/aetiology
Human Genetics (2018)
-
Two mosaic terminal inverted duplications arising post-zygotically: Evidence for possible formation of neo-telomeres
Cell & Chromosome (2008)
-
Dysmorphic features, simplified gyral pattern and 7q11.23 duplication reciprocal to the Williams-Beuren deletion
European Journal of Human Genetics (2008)
-
A 2.3 Mb duplication of chromosome 8q24.3 associated with severe mental retardation and epilepsy detected by standard karyotype
European Journal of Human Genetics (2005)
-
Direct duplication 12p11.21–p13.31 mediated by segmental duplications: a new recurrent rearrangement?
Human Genetics (2005)