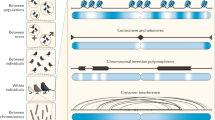
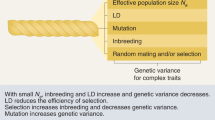
Abstract
Population genetics is central to our understanding of human variation, and by linking medical and evolutionary themes, it enables us to understand the origins and impacts of our genomic differences. Despite current limitations in our knowledge of the locations, sizes and mutational origins of structural variants, our characterization of their population genetics is developing apace, bringing new insights into recent human adaptation, genome biology and disease. We summarize recent dramatic advances, describe the diverse mutational origins of chromosomal rearrangements and argue that their complexity necessitates a re-evaluation of existing population genetic methods.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Feuk, L., Carson, A.R. & Scherer, S.W. Structural variation in the human genome. Nat. Rev. Genet. 7, 85–97 (2006).
Jobling, M.A., Hurles, M.E. & Tyler-Smith, C. Human Evolutionary Genetics: Origins, Peoples and Disease (Garland Science, New York, 2004).
Flint, J. et al. High frequencies of alpha-thalassaemia are the result of natural selection by malaria. Nature 321, 744–750 (1986).
IHMC. A haplotype map of the human genome. Nature 437, 1299–1320 (2005).
Conrad, D.F. et al. A worldwide survey of haplotype variation and linkage disequilibrium in the human genome. Nat. Genet. 38, 1251–1260 (2006).
Bowcock, A.M. et al. High resolution of human evolutionary trees with polymorphic microsatellites. Nature 368, 455–457 (1994).
Armour, J.A.L. et al. Minisatellite diversity supports a recent African origin for modern humans. Nat. Genet. 13, 154–160 (1996).
Myers, S., Bottolo, L., Freeman, C., McVean, G. & Donnelly, P. A fine-scale map of recombination rates and hotspots across the human genome. Science 310, 321–324 (2005).
Sabeti, P.C. et al. Positive natural selection in the human lineage. Science 312, 1614–1620 (2006).
Aitman, T.J. et al. Copy number polymorphism in Fcgr3 predisposes to glomerulonephritis in rats and humans. Nature 439, 851–855 (2006).
Redon, R. et al. Global variation in copy number in the human genome. Nature 444, 444–454 (2006).
Repping, S. et al. High mutation rates have driven extensive structural polymorphism among human Y chromosomes. Nat. Genet. 38, 463–467 (2006).
Schmutz, J. et al. The DNA sequence and comparative analysis of human chromosome 5. Nature 431, 268–274 (2004).
Fernandes, S. et al. A large AZFc deletion removes DAZ3/DAZ4 and nearby genes from men in Y haplogroup N. Am. J. Hum. Genet. 74, 180–187 (2004).
Locke, D.P. et al. Linkage disequilibrium and heritability of copy-number polymorphisms within duplicated regions of the human genome. Am. J. Hum. Genet. 79, 275–290 (2006).
Fiegler, H. et al. Accurate and reliable high-throughput detection of copy number variation in the human genome. Genome Res. 16, 1566–1574 (2006).
Sharp, A.J. et al. Segmental duplications and copy-number variation in the human genome. Am. J. Hum. Genet. 77, 78–88 (2005).
Conrad, D.F., Andrews, T.D., Carter, N.P., Hurles, M.E. & Pritchard, J.K. A high-resolution survey of deletion polymorphism in the human genome. Nat. Genet. 38, 75–81 (2006).
McCarroll, S.A. et al. Common deletion polymorphisms in the human genome. Nat. Genet. 38, 86–92 (2006).
Iafrate, A.J. et al. Detection of large-scale variation in the human genome. Nat. Genet. 36, 949–951 (2004).
Sebat, J. et al. Large-scale copy number polymorphism in the human genome. Science 305, 525–528 (2004).
Chimpanzee Sequencing and Analysis Consortium. Initial sequence of the chimpanzee genome and comparison with the human genome. Nature 437, 69–87 (2005).
Mills, R.E. et al. An initial map of insertion and deletion (INDEL) variation in the human genome. Genome Res. 16, 1182–1190 (2006).
Weber, J.L. et al. Human diallelic insertion/deletion polymorphisms. Am. J. Hum. Genet. 71, 854–862 (2002).
Warburton, D. De novo balanced chromosome rearrangements and extra marker chromosomes identified at prenatal diagnosis: clinical significance and distribution of breakpoints. Am. J. Hum. Genet. 49, 995–1013 (1991).
Linardopoulou, E.V. et al. Human subtelomeres are hot spots of interchromosomal recombination and segmental duplication. Nature 437, 94–100 (2005).
Tuzun, E. et al. Fine-scale structural variation of the human genome. Nat. Genet. 37, 727–732 (2005).
Feuk, L. et al. Discovery of human inversion polymorphisms by comparative analysis of human and chimpanzee DNA sequence assemblies. PLoS Genet 1, e56 (2005).
Khaja, R. et al. Genome assembly comparison identifies structural variants in the human genome. Nat. Genet. 38, 1413–1418 (2006).
Newman, T.L. et al. High-throughput genotyping of intermediate-size structural variation. Hum. Mol. Genet. 15, 1159–1167 (2006).
Perry, G.H. et al. Hotspots for copy number variation in chimpanzees and humans. Proc. Natl. Acad. Sci. USA 103, 8006–8011 (2006).
Jobling, M.A. et al. Recurrent duplication and deletion polymorphisms on the long arm of the Y chromosome in normal males. Hum. Mol. Genet. 5, 1767–1775 (1996).
Clark, A.G., Hubisz, M.J., Bustamante, C.D., Williamson, S.H. & Nielsen, R. Ascertainment bias in studies of human genome-wide polymorphism. Genome Res. 15, 1496–1502 (2005).
Nielsen, R. & Signorovitch, J. Correcting for ascertainment biases when analyzing SNP data: applications to the estimation of linkage disequilibrium. Theor. Popul. Biol. 63, 245–255 (2003).
Stankiewicz, P. & Lupski, J.R. Genome architecture, rearrangements and genomic disorders. Trends Genet. 18, 74–82 (2002).
Shaffer, L.G. & Lupski, J.R. Molecular mechanisms for constitutional chromosomal rearrangements in humans. Annu. Rev. Genet. 34, 297–329 (2000).
Bacolla, A. et al. Breakpoints of gross deletions coincide with non-B DNA conformations. Proc. Natl. Acad. Sci. USA 101, 14162–14167 (2004).
Kurahashi, H. & Emanuel, B.S. Unexpectedly high rate of de novo constitutional t(11;22) translocations in sperm from normal males. Nat. Genet. 29, 139–140 (2001).
Johnson, M.E. et al. Recurrent duplication-driven transposition of DNA during hominoid evolution. Proc. Natl. Acad. Sci. USA 103, 17626–17631 (2006).
Cheng, Z. et al. A genome-wide comparison of recent chimpanzee and human segmental duplications. Nature 437, 88–93 (2005).
Wong, Z., Royle, N.J. & Jeffreys, A.J. A novel human DNA polymorphism resulting from transfer of DNA from chromosome 6 to chromosome 16. Genomics 7, 222–234 (1990).
Balakirev, E.S. & Ayala, F.J. Pseudogenes: are they “junk” or functional DNA? Annu. Rev. Genet. 37, 123–151 (2003).
Kimura, M. The rate of molecular evolution considered from the standpoint of population genetics. Proc. Natl. Acad. Sci. USA 63, 1181–1188 (1969).
Kimura, M. & Crow, J.F. The number of alleles that can be maintained in a finite population. Genetics 49, 725–738 (1964).
Ohta, T. & Kimura, M. A model of mutation appropriate to estimate the number of electrophoretically detectable molecules in a finite population. Genet. Res. 22, 201–204 (1973).
Valdes, A.M., Slatkin, M. & Freimer, N.B. Allele frequencies at microsatellite loci: the stepwise mutation model revisited. Genetics 133, 737–749 (1993).
Bayes, M., Magano, L.F., Rivera, N., Flores, R. & Perez Jurado, L.A. Mutational mechanisms of Williams-Beuren syndrome deletions. Am. J. Hum. Genet. 73, 131–151 (2003).
Han, L.L., Keller, M.P., Navidi, W., Chance, P.F. & Arnheim, N. Unequal exchange at the Charcot-Marie-Tooth disease type 1A recombination hot-spot is not elevated above the genome average rate. Hum. Mol. Genet. 9, 1881–1889 (2000).
Voight, B.F. et al. Interrogating multiple aspects of variation in a full resequencing data set to infer human population size changes. Proc. Natl. Acad. Sci. USA 102, 18508–18513 (2005).
McVean, G.A. et al. The fine-scale structure of recombination rate variation in the human genome. Science 304, 581–584 (2004).
Li, N. & Stephens, M. Modeling linkage disequilibrium and identifying recombination hotspots using single-nucleotide polymorphism data. Genetics 165, 2213–2233 (2003).
Andolfatto, P. & Nordborg, M. The effect of gene conversion on intralocus associations. Genetics 148, 1397–1399 (1998).
Hudson, R.R., Bailey, K., Skarecky, D., Kwiatowski, J. & Ayala, F.J. Evidence for positive selection in the superoxide dismutase (Sod) region of Drosophila melanogaster. Genetics 136, 1329–1340 (1994).
Sabeti, P.C. et al. Detecting recent positive selection in the human genome from haplotype structure. Nature 419, 832–837 (2002).
Zondervan, K.T. & Cardon, L.R. The complex interplay among factors that influence allelic association. Nat. Rev. Genet. 5, 89–100 (2004).
Fredman, D. et al. Complex SNP-related sequence variation in segmental genome duplications. Nat. Genet. 36, 861–866 (2004).
Pritchard, J.K. Are rare variants responsible for susceptibility to complex diseases? Am. J. Hum. Genet. 69, 124–137 (2001).
Wright, S. The genetical structure of populations. Ann. Eugen. 15, 323–354 (1951).
Nei, M. Analysis of gene diversity in subdivided populations. Proc. Natl. Acad. Sci. USA 70, 3321–3323 (1973).
Watkins, W.S. et al. Genetic variation among world populations: inferences from 100 Alu insertion polymorphisms. Genome Res. 13, 1607–1618 (2003).
Barbujani, G., Magagni, A., Minch, E. & Cavalli-Sforza, L.L. An apportionment of human DNA diversity. Proc. Natl. Acad. Sci. USA 94, 4516–4519 (1997).
Pritchard, J.K., Stephens, M. & Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 155, 945–959 (2000).
Gonzalez, E. et al. The influence of CCL3L1 gene-containing segmental duplications on HIV-1/AIDS susceptibility. Science 307, 1434–1440 (2005).
Stefansson, H. et al. A common inversion under selection in Europeans. Nat. Genet. 37, 129–137 (2005).
Nguyen, D.Q., Webber, C. & Ponting, C.P. Bias of selection on human copy-number variants. PLoS Genet. 2, e20 (2006).
Hinds, D.A., Kloek, A.P., Jen, M., Chen, X. & Frazer, K.A. Common deletions and SNPs are in linkage disequilibrium in the human genome. Nat. Genet. 38, 82–85 (2006).
Brewer, C., Holloway, S., Zawalnyski, P., Schinzel, A. & FitzPatrick, D. A chromosomal duplication map of malformations: regions of suspected haplo- and triplolethality–and tolerance of segmental aneuploidy–in humans. Am. J. Hum. Genet. 64, 1702–1708 (1999).
Johnson, M.E. et al. Positive selection of a gene family during the emergence of humans and African apes. Nature 413, 514–519 (2001).
Popesco, M.C. et al. Human lineage-specific amplification, selection, and neuronal expression of DUF1220 domains. Science 313, 1304–1307 (2006).
Voight, B.F., Kudaravalli, S., Wen, X. & Pritchard, J.K. A map of recent positive selection in the human genome. PLoS Biol. 4, e72 (2006).
Bailey, J.A. et al. Recent segmental duplications in the human genome. Science 297, 1003–1007 (2002).
Przeworski, M., Hudson, R.R. & Di Rienzo, A. Adjusting the focus on human variation. Trends Genet. 16, 296–302 (2000).
Bubb, K.L. et al. Scan of human genome reveals no new Loci under ancient balancing selection. Genetics 173, 2165–2177 (2006).
Turner, D.J. et al. Assaying chromosomal inversions by single-molecule haplotyping. Nat. Methods 3, 439–445 (2006).
Slatkin, M. & Rannala, B. Estimating allele age. Annu. Rev. Genomics Hum. Genet. 1, 225–249 (2000).
Acknowledgements
The authors are grateful to G. Coop and C. Tyler-Smith for their comments on an earlier manuscript and to D. Andrews for data processing.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Heritability of a multiallelic CNV. (PDF 40 kb)
Supplementary Fig. 2
Haplotype patterns within a common polymorphic inversion on 8p23. (PDF 12 kb)
Rights and permissions
About this article
Cite this article
Conrad, D., Hurles, M. The population genetics of structural variation. Nat Genet 39 (Suppl 7), S30–S36 (2007). https://doi.org/10.1038/ng2042
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng2042
This article is cited by
-
Decoding the mechanism of hypertension through multiomics profiling
Journal of Human Hypertension (2022)
-
Algorithm for sample availability prediction in a hospital-based epidemiological study spreadsheet-based sample availability calculator
Scientific Reports (2022)
-
Megabase-scale presence-absence variation with Tripsacum origin was under selection during maize domestication and adaptation
Genome Biology (2021)
-
Detection of copy number variants in African goats using whole genome sequence data
BMC Genomics (2021)
-
Analysis of structural variants in four African cichlids highlights an association with developmental and immune related genes
BMC Evolutionary Biology (2020)