Abstract
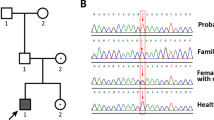
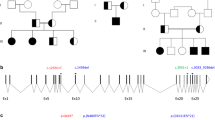
ADRENAL hypoplasia congenita (AHC) is an X-linked disorder characterized by primary adrenal insufficiency1,2. Hypogonadotropic hypogonadism (HHG) is frequently associated with this disorder but is thought not to be caused by the low adrenal androgen levels due to adrenal hypoplasia3,4. It is uncertain whether there are two distinct yet physically linked genes responsible for AHC and HHG or a single gene responsible for both diseases. AHC can occur as a part of a contiguous deletion syndrome together with Duchenne muscular dystrophy (DMD) and/or glycerol kinase deficiency (GKD). From the analysis of deletions, the following gene order has been deduced: Xpter-AHC-GKD-DMD-cen5,6. An AHC critical region of 200–500 kilobases has been defined by physical mapping7,8 and partially overlaps with a 160-kilobase dosage-sensitive sex (DSS) reversal critical region9. The DAX-1 (DSS-AHC critical region on the X, gene 1) gene was isolated and found to encode a new member of the nuclear hormone receptor family10. Here we report that DAX-1 is deleted in 14 patients and point mutations were found in the coding region in DNA from 12 unrelated individuals. All AHC patients over 14 years old and with only point mutations in DAX-1 were also diagnosed with HHG, confirming that the DAX-1 gene is responsible for both X-linked AHC and HHG. But in four sporadic cases and a single familial case, no point mutations were found, suggesting genetic heterogeneity or differential expression of DAX-1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kelch, R. P. et al. Pediatr. Adolesc. Endocrin. 13, 156–161 (1984).
Kleczkowska, A., Kubien, E., Dmoch, E., Fryns, J. P. & Van den Berghe, H. Genet. Counseling 1, 241–249 (1991).
Cohen, H. N., Hay, I. D., Beastall, G. H. & Thomson, J. A. Lancet II, 1471–1472 (1982).
Kikuchi, K., Kaji, M., Mikawa, H., Shimegatsu, S. & Sudo, M. Acta Endocrin. 114, 153–160 (1987).
Francke, U. et al. Am. J. hum. Genet. 40, 212–227 (1987).
Goonewardena, P., Dahl, N., Ritzén, M., Van Ommen, G. J. B. & Petterson, U. Clin. Genet. 35, 5–12 (1989).
Walker, A. P. et al. Hum. molec. Genet. 1, 579–585 (1992).
Worley, K. C. et al. Genomics 16, 407–416 (1993).
Bardoni, B. et al. Nature Genet. 7, 497–501 (1994).
Zanaria, E. et al. Nature 372, 635–641 (1994).
Hultman, T., Stahl, S., Hornes, E. & Uhlén, M. Nucleic Acids Res. 17, 4937–4946 (1989).
Baserga, S. L. & Benz, E. J. Proc. natn. Acad. Sci. U.S.A. 85, 2056–2060 (1988).
Naylor, J. A., Green, P. M., Rizza, C. R. & Giannelli, F. Hum. molec. Genet. 2, 11–17 (1993).
Lakich, D., Kazazian, H. H., Antonarakis, S. E. & Gitschier, J. Nature Genet. 5, 236–241 (1993).
Chelly, J., Concordet, J., Kaplan, J. C. & Kahn, A. Proc. natn. Acad. Sci. U.S.A. 86, 2617–2621 (1990).
Sizonenko, P. C. Am. J. Dis. Child. 132, 704–806 (1978).
Luo, X., Ikeda, Y. & Parker, K. L. Cell 77, 481–490 (1994).
Nizetic, D. et al. Proc. natn. Acad. Sci. U.S.A. 88, 3233–3237 (1991).
Baxendale, S. et al. Nature Genet. 4, 181–186 (1993).
Rackwitz, H. R. Gene 40, 259–266 (1985).
Sambrook, J., Fritsh, E. F. & Maniatis, T. Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory Press, New York, 1989).
Zehetner, G. & Lehrach, H. Nucleic Acids Res. 14, 335–349 (1986).
Fairweather, N. et al. Hum. molec. Genet. 3, 29–34 (1994).
Walker, A. P., Muscatelli, F. & Monaco, A. P. Hum. molec. Genet. 2, 107–114 (1993).
Yates, J. R. W. et al. Cytogenet. Cell Genet. 46, 723–724 (1987).
Matfin, G. et al. Clin. Endocrin. 40, 807–808 (1994).
Marhlens, F. et al. Hum. Genet. 77, 379–383 (1987).
Récan, D. et al. J. clin. Invest. 89, 712–716 (1992).
Chelly, J. et al. Hum. Genet. 78, 222–227 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Muscatelli, F., Strom, T., Walker, A. et al. Mutations in the DAX-1 gene give rise to both X-linked adrenal hypoplasia congenita and hypogonadotropic hypogonadism. Nature 372, 672–676 (1994). https://doi.org/10.1038/372672a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/372672a0
This article is cited by
-
Clinical and genetic characteristics of 42 Chinese paediatric patients with X-linked adrenal hypoplasia congenita
Orphanet Journal of Rare Diseases (2023)
-
Leydig Cell–Specific DAX1-Deleted Mice Has Higher Testosterone Level in the Testis During Pubertal Development
Reproductive Sciences (2022)
-
X-linked congenital adrenal hypoplasia: a case presentation
BMC Endocrine Disorders (2021)
-
Adrenal cortex renewal in health and disease
Nature Reviews Endocrinology (2021)
-
Genetics of congenital hypogonadotropic hypogonadism: peculiarities and phenotype of an oligogenic disease
Human Genetics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.