Abstract
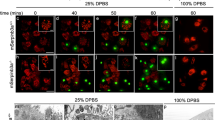
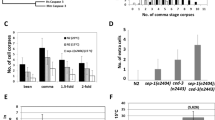
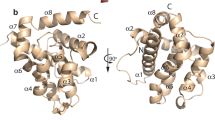
During programmed cell death, cell corpses are rapidly engulfed1. This engulfment process involves the recognition and subsequent phagocytosis of cell corpses by engulfing cells1,2,3,4. How cell corpses are engulfed is largely unknown. Here we report that ced-5, a gene that is required for cell-corpse engulfment in the nematode Caenorhabditis elegans5, encodes a protein that is similar to the human protein DOCK180 and the Drosophila melanogaster protein Myoblast City (MBC), both of which have been implicated in the extension of cell surfaces6. ced-5 mutants are defective not only in the engulfment of cell corpses but also in the migrations of two specific gonadal cells, the distal tip cells. The expression of human DOCK180 in C. elegans rescued the cell-migration defect of a ced-5 mutant. We present evidence that ced-5 functions in engulfing cells during the engulfment of cell corpses. We suggest that ced-5 acts in the extension of the surface of an engulfing cell around a dying cell during programmed cell death. We name this new family of proteins that function in the extension of cell surfaces the CDM (for CED-5, DOCK180 and MBC) family.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ellis, R. E., Yuan, J. & Horvitz, H. R. Mechanisms and functions of cell death. Annu. Rev. Cell Biol. 7, 663–698 (1991).
Savill, J., Fadok, V., Henson, P. & Haslett, C. Phagocyte recognition of cells undergoing apoptosis. Immunol. Today 14, 131–136 (1993).
Hart, S. P., Haslett, C. & Dransfield, I. Recognition of apoptotic cells by phagocytes. Experientia 52, 950–956 (1996).
Savill, J. Apoptosis in resolution of inflammation. J. Leuk. Biol. 61, 375–380 (1997).
Ellis, R., Jacobson, D. M. & Horvitz, H. R. Genes required for the engulfment of cell corpses during programmed cell death in C. elegans. Genetics 129, 79–94 (1991).
Hesegawa, H. et al. DOCK180, a major CRK-binding protein, alters cell morphology upon translocation to the cell membrane. Mol. Cell. Biol. 16, 1770–1776 (1996).
Hedgecock, E., Sulston, J. & Thomson, J. N. Mutations affecting programmed cell deaths in the nematode Caenorhabditis elegans. Science 220, 1277–1279 (1983).
Kimble, J. Alterations in cell lineage following laser ablation of cells in the somatic gonad of Caenorhabditis elegans. Dev. Biol. 87, 286–300 (1981).
Hedgecock, E. M., Culotti, J. G., Hall, D. H. & Stern, B. D. Genetics of cell and axon migrations in Caenorhabditis elegans. Development 100, 365–382 (1987).
Coulson, A., Sulston, J., Brenner, S. & Karn, J. Toward a physical map of the genome of the nematode Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 83, 7821–7825 (1986).
Way, J. C. & Chalfie, M. mec-3, a homeobox-containing gene that specifies differentiation of the touch receptor neurons in C. elegans. Cell 54, 5–16 (1988).
Krause, M. & Hirsh, D. Atrans-spliced leader sequence on actin mRNA in C. elegans. Cell 49, 753–761 (1987).
Stringham, E. G., Dixon, D. K., Jones, D. & Candido, E. P. Temporal and spatial expression patterns of the small heat shock (hsp16) genes in transgenic Caenorhabditis elegans. Mol. Biol. Cell 3, 221–233 (1992).
Erickson, M., Galletta, B. J. & Abmayr, S. M. Drosophila myoblast city encodes a conserved protein that is essential for myoblast fusion, dorsal closure, and cytoskeletal organization. J. Cell Biol. 138, 589–603 (1997).
Nagase, T. et al. Prediction of the coding sequences of unidentified human genes. VI. The coding sequences of 80 new genes (KIAA0201-KIAA0280) deduced by analysis of cDNA clones from cell line KG-1 and brain. DNA Res. 3, 321–329 (1996).
Mayer, B. J., Hamaguchi, M. & Hanafusa, H. Anovel viral oncogene with structural similarity to phospholipase C. Nature 332, 272–275 (1988).
Clark, E. A. & Brugge, J. S. Integrins and signal transduction pathways: the road taken. Science 268, 233–239 (1995).
Savill, J., Dransfield, I., Hogg, N. & Haslett, C. Vitronectin receptor-mediated phagocytosis of cells undergoing apoptosis. Nature 343, 170–173 (1990).
Rushton, E., Drysdale, R., Abmayr, S. M., Michelson, A. M. & Bate, M. Mutations in a novel gene, myoblast city, provide evidence in support of the founder cell hypothesis for Drosophila muscle development. Development 121, 1979–1988 (1995).
Chalfie, M., Tu, Y., Euskirchen, G., Ward, W. W. & Prasher, D. C. Green fluorescent protein as a marker for gene expression. Science 263, 802–805 (1994).
Fire, A., Harrison, S. W. & Dixon, D. Amodular set of lacZ fusion vectors for studying gene expression in Caenorhabditis elegans. Gene 93, 189–198 (1990).
Robertson, A. G. & Thomson, J. N. Morphology of programmed cell death in the ventral nerve cord of C. elegans larvae. J. Embryo. Exp. Morph. 67, 89–100 (1982).
Yuan, J., Shaham, S., Ledoux, S., Ellis, H. M. & Horvitz, H. R. The C. elegans cell death gene ced-3 encodes a protein similar to mammalian interleukin-1β-converting enzyme. Cell 75, 641–652 (1993).
Hengartner, M. O. & Horvitz, H. R. C. elegans cell survival gene ced-9 encodes a functional homolog of the mammalian proto-oncogene bcl-2. Cell 76, 665–676 (1994).
Zou, H., Henzel, W. J., Liu, X., Lutschg, A. & Wang, X. Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 90, 405–413 (1997).
Mello, C. C., Kramer, J. M., Stinchcomb, D. & Ambros, V. Efficient gene transfer in C. elegans: Extrachromosomal maintenance and integration of transforming sequences. EMBO J. 10, 3959–3970 (1992).
Bloom, L. & Horvitz, H. R. The Caenorhabditis elegans gene unc-76 and its human homologs define a new gene family involved in axonal outgrowth and fasciculation. Proc. Natl Acad. Sci. USA 94, 3414–3419 (1997).
Barstead, R. J. & Waterston, R. H. The basal component of the nematode dense-body is vinculin. J. Biol. Chem. 264, 10177–10185 (1989).
Okkema, P. G. & Fire, A. The Caenorhabditis elegans NK-2 class homeoprotein CEH-22 is involved in combinatorial activation of gene expression in pharyngeal muscle. Development 120, 2175–2186 (1994).
Acknowledgements
We thank members of H.R.H.'s laboratory and C.-L. Wei for their comments, D. Hall and E. Hedgecock for sharing unpublished results regarding DTC migration, R. Barstead and P. Okkema for cDNA libraries, A Coulson for cosmids, H. Hasegawa and M. Matsuda for the DOCK180 cDNA clone, E. James for determining DNA sequences of the RACE product, S. Glass for the ced-5 alleles n2098 and n2099 and J. Harris and C. Kenyon for mu57. H.R.H. is an Investigator of the Howard Hughes Medical Institute. The accession number for the ced-5 sequence is AF038576.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, YC., Horvitz, H. C. elegans phagocytosis and cell-migration protein CED-5 is similar to human DOCK180. Nature 392, 501–504 (1998). https://doi.org/10.1038/33163
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/33163
This article is cited by
-
Biasing the conformation of ELMO2 reveals that myoblast fusion can be exploited to improve muscle regeneration
Nature Communications (2022)
-
Replication stress promotes cell elimination by extrusion
Nature (2021)
-
The cisd gene family regulates physiological germline apoptosis through ced-13 and the canonical cell death pathway in Caenorhabditis elegans
Cell Death & Differentiation (2019)
-
EFF-1 fusogen promotes phagosome sealing during cell process clearance in Caenorhabditis elegans
Nature Cell Biology (2018)
-
Engulfing cells promote neuronal regeneration and remove neuronal debris through distinct biochemical functions of CED-1
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.