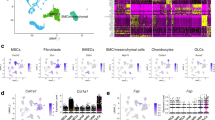
Abstract
Although the perinatal lethal form1 of osteogenesis imperfecta (OI type II) occasionally results from large rearrangements within the genes encoding type I collagen2–5, most mutations are far more subtle. The complexity of the human collagen genes6 precludes cloning and sequencing each gene from every patient, and we have therefore developed an approach to localizing mutations at the protein level. We report here that cells cultured from 15 infants with OI type II synthesized both normal type I procollagen and a form that was unstable, poorly secreted and excessively modified. Abnormal procollagen from different strains was overmodified to different extents. The patterns of overmodification we observed are best explained by mutations that disrupt the Gly-X-Y sequence of proα chains, and thus alter the rate of propagation of triple helix from COOH-terminus to NH2-terminus7. As a consequence, a given mutation allows overmodification of all three chains in a molecule NH2-terminal to its position in the triple helix.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sillence, D. O., Senn, A. S. & Danks, D. M. J. med. Genet. 16, 101–116 (1979).
Hollister, D. W., Byers, P. H. & Holbrook, K. A. Adv. hum. Genet. 12, 1–83 (1982).
Byers, P. H. & Bonadio, J. F. in International Medical Reviews, Pediatrics 5, Genetic and Metabolic Disease (eds Scriver, C. R. & Lloyd, J.) (Butterworths, London, in the press).
Prockop, D. J. & Kivirikko, K. New Engl. J. Med. 311, 376–386 (1984).
Prockop, D. J. J. clin. Invest. 75, 783–787 (1985).
Tate, V., Finer, M., Boedtker, H. & Doty, P. Cold Spring Harb. Symp. quant. Biol. 47, 1039–1049 (1982).
Bachinger, H. P., Fessler, L. I., Timpl, R. & Fessler, J. H. J. biol. Chem. 256, 13193–13199 (1981).
Bernstein, P., Kang, A. H. & Piez, K. A. Biochemistry 5, 3803–3812 (1966).
Kivirikko, K. I. & Myllyla, R. Meth. Enzym. 82, 245–304 (1982).
Barsh, G. S. & Byers, P. H. Proc. natn. Acad. Sci. U.S.A. 78, 5142–5146 (1981).
Barsh, G. S., Roush, C. L., Bonadio, J., Byers, P. H. & Gelinas, R. E. Proc, natn. Acad Sci U.S.A. 82, 2870–2874 (1985).
Williams, C. J. & Prockop, D. J. J. biol. Chem. 258, 5915–5921 (1983).
Chu, M.-L. et al. Nature 304, 78–80 (1983).
de Wet, W. J., Pihlajaniemi, T., Myers, J., Kelly, T. E. & Prockop, D. J. J. biol. Chem. 258, 7721–7728 (1983).
Bonadio, J., Holbrook, K. A., Gelinas, R. E., Jacob, J. & Byers, P. H. J. biol. Chem. 260, 1734–1742 (1985).
Stricklin, G. P., Eisen, F. Z., Bauer, E. A. & Jeffrey, J. J. Biochemistry 17, 2331–2337 (1978).
Prockop, D. J., Kivirikko, K. I., Tuderman, L. & Guzman, N. A. New Engl. J. Med. 301, 13–23 (1979).
Steinmann, B. et al. J. biol. Chem. 259, 11129–11138 (1984).
Trelstad, R. L., Rubin, D. & Gross, J. Lab. Invest. 36, 501–508 (1977).
Kirsch, E. et al. Eur. J. clin. Invest. 11, 39–47 (1981).
Bateman, J. F., Mascara, T., Chan, D. & Cole, W. G. Biochem. J. 217, 103–115 (1984).
Byers, P. H., et al. Am. J. hum. Genet. 35, 39A (1983).
Dixon, L. A. et al. Proc. natn. Acad. Sci. U.S.A. 81, 4525–4528 (1984).
Pihlajaniemi, T. et al. J. biol. Chem. 259, 12941–12944 (1984).
Sillence, D. O., Barlow, K. K., Garber, A. P., Hall, J. G. & Rimoin, D. Am. J. med. Genet. 17, 407–423 (1984).
Laemmli, U. K. Nature 227, 680–685 (1970).
Barsh, G. S., Peterson, K. E. & Byers, P. H. Collagen and Related Research 1, 543–548 (1981).
Bruckner, P., Eikenberry, E. F. & Prockop, D. J. Eur. J. Biochem. 118, 607–613 (1981).
Chu, M.-L., Gargiulo, Y., Williams, C. & Ramirez, F. J. biol. Chem. 260, 691–694 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bonadio, J., Byers, P. Subtle structural alterations in the chains of type I procollagen produce osteogenesis imperfecta type II. Nature 316, 363–366 (1985). https://doi.org/10.1038/316363a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/316363a0
This article is cited by
-
Distinct functions of the laminin β LN domain and collagen IV during cardiac extracellular matrix formation and stabilization of alary muscle attachments revealed by EMS mutagenesis in Drosophila
BMC Developmental Biology (2014)
-
Mutation screening by a combination of biotin-SSCP and direct sequencing
Human Genetics (1994)
-
Embryonic lethality caused by mutations in basement membrane collagen of C. elegans
Nature (1991)
-
Phenotypic variability and abnormal type I collagen unstable at body temperature in a family with mild dominant osteogenesis imperfecta
Journal of Inherited Metabolic Disease (1991)
-
Cysteine in the triple helical domain of the pro?2(I) chain of type-I collagen in nonlethal forms of osteogenesis imperfecta
Human Genetics (1991)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.