Abstract
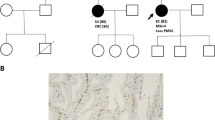
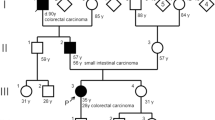
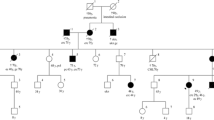
Turcot syndrome is a rare, inherited disease predisposing of tumours in the central nerve system and in the colorectal system. This report describes a Turcot patient with an extraordinary clinical history. The patient is still alive at the age of 43. She was operated at the age of 10 by brain tumour and at the age of 16 by colorectal cancer. She has since then been treated for multiple cancers (gastrointestinal, endometrial, basal cell carcinomas), and removal of adenomatous polyps at several occasions. The aim of this work was to investigate if there was any specific genotype that explains her remarkable clinical history. Microsatellite instability and immunohistochemistry analysis for four DNA mismatch repair proteins were performed. DNA mutation analysis was done for genes involved in polyposis and mismatch repair by denaturing high performance liquid chromatography and sequencing. cDNA analysis was carried out for the mismatch repair gene PMS2. The patients genotype was found to be a homozygous splice site mutation in the PMS2 gene, c.989-1G<T, which resulted in two abnormal transcripts, not one as expected. The patient’s long time survival may in part be explained by meticulous follow up by health care professionals. The other importing factor is probably the nature of here genotype. cDNA analysis showed that the homozygous mutation led to two abnormal transcripts, of which one is perhaps less detrimental. Thus cDNA analysis is of prime importance for the full evaluation of the effect of putative splicing mutations.




Similar content being viewed by others
Abbreviations
- APC:
-
Adenomatous polyposis coli
- CNS:
-
Central nerve system
- HNPCC:
-
Hereditary nonpolyposis colorectal cancer
- MUTYH:
-
MutY E. coli homologue
References
Turcot J, Despres JP, St Pierre F (1959) Malignant tumors of the central nervous system associated with familial polyposis of the colon: report of two cases. Dis Colon Rectum 2:465–468. doi:10.1007/BF02616938
Hamilton SR, Liu B, Parsons RE et al (1995) The molecular basis of Turcot’s syndrome. N Engl J Med 332:839–847. doi:10.1056/NEJM199503303321302
Paraf F, Jothy S, Van Meir EG (1997) Brain tumor-polyposis syndrome: two genetic diseases? J Clin Oncol 15:2744–2758
Trimbath JD, Petersen GM, Erdman SH et al (2001) Cafe-au-lait spots and early onset colorectal neoplasia: a variant of HNPCC? Fam Cancer 1:101–105. doi:10.1023/A:1013881832014
Loukola A, Eklin K, Laiho P et al (2001) Microsatellite marker analysis in screening for hereditary nonpolyposis colorectal cancer (HNPCC). Cancer Res 61:4545–4549
Aaltonen LA, Salovaara R, Kristo P et al (1998) Incidence of hereditary nonpolyposis colorectal cancer and the feasibility of molecular screening for the disease. N Engl J Med 338:1481–1487. doi:10.1056/NEJM199805213382101
Groden J, Thliveris A, Samowitz W et al (1991) Identification and characterization of the familial adenomatous polyposis coli gene. Cell 66:589–600. doi:10.1016/0092-8674(81)90021-0
Jones S, Emmerson P, Maynard J et al (2002) Biallelic germline mutations in MYH predispose to multiple colorectal adenoma and somatic G:C–>T:A mutations. Hum Mol Genet 11:2961–2967. doi:10.1093/hmg/11.23.2961
Weber TK, Conlon W, Petrelli NJ et al (1997) Genomic DNA-based hMSH2 and hMLH1 mutation screening in 32 Eastern United States hereditary nonpolyposis colorectal cancer pedigrees. Cancer Res 57:3798–3803
Nicolaides NC, Carter KC, Shell BK et al (1995) Genomic organization of the human PMS2 gene family. Genomics 30:195–206. doi:10.1006/geno.1995.9885
De Vos M, Hayward BE, Picton S et al (2004) Novel PMS2 pseudogenes can conceal recessive mutations causing a distinctive childhood cancer syndrome. Am J Hum Genet 74:954–964. doi:10.1086/420796
Al-Tassan N, Chmiel NH, Maynard J et al (2002) Inherited variants of MYH associated with somatic G:C–>T:A mutations in colorectal tumors. Nat Genet 30:227–232. doi:10.1038/ng828
De Rosa M, Fasano C, Panariello L et al (2000) Evidence for a recessive inheritance of Turcot’s syndrome caused by compound heterozygous mutations within the PMS2 gene. Oncogene 19:1719–1723. doi:10.1038/sj.onc.1203447
Nakagawa H, Lockman JC, Frankel WL et al (2004) Mismatch repair gene PMS2: disease-causing germline mutations are frequent in patients whose tumors stain negative for PMS2 protein, but paralogous genes obscure mutation detection and interpretation. Cancer Res 64:4721–4727. doi:10.1158/0008-5472.CAN-03-2879
Agostini M, Tibiletti MG, Lucci-Cordisco E et al (2005) Two PMS2 mutations in a Turcot syndrome family with small bowel cancers. Am J Gastroenterol 100:1886–1891. doi:10.1111/j.1572-0241.2005.50441.x
Gottschling S, Reinhard H, Pagenstecher C et al (2008) Hypothesis: possible role of retinoic acid therapy in patients with biallelic mismatch repair gene defects. Eur J Pediatr 167(2):225–229. doi:10.1007/s00431-007-0474-3
Taylor MD, Perry J, Zlatescu MC et al (1999) The hPMS2 exon 5 mutation and malignant glioma. Case report. J Neurosurg 90:946–950
Clendenning M, Hampel H, LaJeunesse J et al (2006) Long-range PCR facilitates the identification of PMS2-specific mutations. Hum Mutat 27:490–495. doi:10.1002/humu.20318
Hendriks YMC, Jagmohan-Changur S, van der Klift HM et al (2006) Heterozygous mutation in PMS2 cause hereditary nonpolyposis colorectal Carcinoma (Lynch Syndrome). Gastroenterology 130:312–322. doi:10.1053/j.gastro.2005.10.052
Charames GS, Cheng H, Gilpin CA et al (2002) A novel aberrant splice site mutation in the APC gene. J Med Genet 39:754–757. doi:10.1136/jmg.39.10.754
Yu L, Heere-Ress E, Boucher B, et al. (1999) Familial hypercholesterolemia. Acceptor splice site (G–>C) mutation in intron 7 of the LDL-R gene: alternate RNA editing causes exon 8 skipping or a premature stop codon in exon 8. LDL-R(Honduras-1). Atherosclerosis 146:125–131. LDL-R1061(-1) G–>C. doi:10.1016/S0021-9150(99)00109-4
Andreutti-Zaugg C, Scott RJ, Iggo R (1997) Inhibition of nonsense-mediated Messenger RNA decay in clinical samples facilitates detection of human MSH2 mutations with an in vivo fusion protein assay and conventional techniques. Cancer Res 57:3288–3293
Behm-Ansmant I, Kashima I, Rehwinkel J et al (2007) mRNA quality control: an ancient machinery recognizes and degrades mRNAs with nonsense codons. FEBS Lett 15:2845–2853. doi:10.1016/j.febslet.2007.05.027
Ban C, Junop M, Yang W (1999) Transformation of MutL by ATP binding and hydrolysis: a switch in DNA mismatch repair. Cell 97:85–97. doi:10.1016/S0092-8674(00)80717-5
Guarne A, Junop MS, Yang W (2001) Structure and function of the N-terminal 40 kDa fragment of human PMS2: a monomeric GHL ATPase. EMBO J 20:5521–5531. doi:10.1093/emboj/20.19.5521
Truninger K, Menigatti M, Luz J (2005) Immunohistochemical analysis reveals high frequency of PMS2 defects in colorectal cancer. Gastroenterology 128:1160–1171. doi:10.1053/j.gastro.2005.01.056
Worthley DL, Walsh MD, Barker M (2005) Familial mutations in PMS2 can cause autosomal dominant hereditary nonpolyposis colorectal cancer. Gastroenterology 128:1431–1436. doi:10.1053/j.gastro.2005.04.008
Gryfe R, Gallinger S (2005) Germline PMS2 mutations: one hit or two? Gastroenterology 128:1506–1509. doi:10.1053/j.gastro.2005.03.054
Falkenback D, Johansson J, Halvarsson B, Nilbert M (2005) Defective mismatch-repair as a minor tumorigenic pathway in Barrett esophagus-associated adenocarcinoma. Cancer Genet Cytogenet 157:82–86. doi:10.1016/j.cancergencyto.2004.08.003
Rutz HP, de Tribolet N, Calmes JM, Chapuis G (1991) Long-time survival of a patient with glioblastoma and Turcot’s syndrome. Case report. J Neurosurg 74:813–815
De Vos M, Hayward BE, Charlton R (2006) PMS2 mutations in childhood cancer. J Natl Cancer Inst 98:358–361
Acknowledgments
This work was supported in part by the Cancer Foundation at St Olavs University Hospital. We thank L. A. Lavik, B. Gilde, S. Brathaug, K. Bye and E. Svaasand for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sjursen, W., Bjørnevoll, I., Engebretsen, L.F. et al. A homozygote splice site PMS2 mutation as cause of Turcot syndrome gives rise to two different abnormal transcripts. Familial Cancer 8, 179–186 (2009). https://doi.org/10.1007/s10689-008-9225-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10689-008-9225-5