Abstract
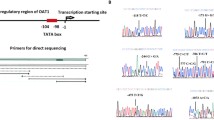
Using a large-scale genotyping analysis of gene-based single nucleotide polymorphisms (SNPs) in Japanese type 2 diabetic patients, we have identified a gene encoding neurocalcin δ (NCALD) as a candidate for a susceptibility gene to diabetic nephropathy; the landmark SNP was found in the 3′ UTR of NCALD (rs1131863: exon 4 +1340 A vs. G, P = 0.00004, odds ratio = 1.59, 95% CI 1.27–1.98). We also discovered two other SNPs in exon 4 of this gene (+999 T/A, +1307 A/G) that showed absolute linkage disequilibrium to the landmark SNP. Subsequent in vitro functional analysis revealed that synthetic mRNA corresponding to the disease susceptible haplotype (exon 4 +1340 G, +1307 G, +999 A) was degraded faster than mRNA corresponding to the major haplotype (exon 4 +1340 A, +1307 A, +999 T), and allelic mRNA expression of the disease susceptibility allele was significantly lower than that of the major allele in normal kidney tissues. In an experiment using a short interfering RNA targeting NCALD, we found that silencing of the NCALD led to a considerable enhancement of cell migration, accompanied by a significant reduction in E-cadherin expression, and by an elevation of α smooth muscle actin expression in cultured renal proximal tubular epithelial cells. We also identified the association of the landmark SNP with the progression of diabetic nephropathy in a 8-year prospective study (A vs. G, P = 0.03, odds ratio = 1.91, 95% CI 1.07–3.42). These results suggest that the NCALD gene is a likely candidate for conferring susceptibility to diabetic nephropathy.




Similar content being viewed by others
Abbreviations
- SNP:
-
Single nucleotide polymorphism
- LD:
-
Linkage disequilibrium
- HWE:
-
Hardy–Weinberg equilibrium
- TEMT:
-
Tubular epithelial mesenchymal-transdifferentiation
- UTR:
-
Untranslated region
References
Akmal M (2001) Hemodialysis in diabetic patients. Am J Kidney Dis 38:S195–S199
Araki S, Haneda M, Sugimoto T, Isono M, Isshiki K, Kashiwagi A, Koya D (2005) Factors associated with frequent remission of microalbuminuria in patients with type 2 diabetes. Diabetes 54:2983–2987
Bedell JA, Korf I, Gish W (2000) MaskerAid: a performance enhancement to RepeatMasker. Bioinformatics 16:1040–1041
Bottinger EP, Bitzer M (2002) TGF-β signaling in renal disease. J Am Soc Nephrol 13:2600–2610
Bowden DW, Colicigno CJ, Langefeld CD, Sale MM, Williams A, Anderson PJ, Rich SS, Freedman BI (2004) A genome scan for diabetic nephropathy in African Americans. Kidney Int 66:1517–1526
Collins AJ, Kasiske B, Herzog C, Chavers B, Foley R, Gilbertson D, Grimm R, Liu J, Louis T, Manning W, McBean M, Murray A, St Peter W, Xue J, Fan Q, Guo H, Li Q, Li S, Qiu Y, Li S, Roberts T, Skeans M, Snyder J, Solid C, Wang C, Weinhandl E, Zhang R, Arko C, Chen SC, Dalleska F, Daniels F, Dunning S, Ebben J, Frazier E, Hanzlik C, Johnson R, Sheets D, Wang X, Forrest B, Berrini D, Constantini E, Everson S, Eggers P, Agodoa L (2006) Excerpts from the United States Renal Data System 2006 annual data report: atlas of end-stage renal disease in the United States. Am J Kidney Dis 49:S1–S296
Collins FS, Guyer MS, Chakravarti A (1997) Variations on a theme: cataloging human DNA sequence variation. Science 278:1580–1581
Devlin B, Risch N (1995) A comparison of linkage disequilibrium measures for fine-scale mapping. Genomics 29:311–322
Faulkner JL, Szcykalski LM, Springer F, Barnes JL (2005) Origin of interstitial fibroblasts in an accelerated model of angiotensin II-induced renal fibrosis. Am J Pathol 167:1193–1205
Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O (2003) Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 348:383–393
Ha H, Lee HB (2003) Reactive oxygen species and matrix remodeling in diabetic kidney. J Am Soc Nephrol 14:S246–S249
Haga H, Yamada R, Ohnishi Y, Nakamura Y, Tanaka T (2002) Gene-based SNP discovery as part of the Japanese Millennium Genome Project: identification of 190,562 genetic variation in the human genome. J Hum Genet 47:605–610
Hidaka H, Okazaki K (1993) Neurocalcin family. A novel calcium-binding protein abundant in bovine central nervous system. Neurosci Res 16:73–77
Hirakawa M, Tanaka T, Hashimoto Y, Kuroda M, Takagi T, Nakamura Y (2002) JSNP a database of common gene variations in the Japanese population. Nucleic Acids Res 30:158–162
Imperatore G, Hanson RL, Pettitt DJ, Kobes S, Bennett PH, Knowler WC (1998) Sib-pair linkage analysis for susceptibility genes for microvascular complications among Pima Indians with type 2 diabetes. Pima Diabetes Genes Group. Diabetes 47:821–830
International Human Genome Sequencing Consortium (2004) Finishing the euchromatic sequence of the human genome. Nature 431:931–945
Kalluri R, Neilson E (2003) Epithelial–mesenchymal transition and its implication for fibrosis. J Clin Invest 112:1776–1784
Krolewski AS, Warram JH, Rand LI, Kahn CR (1987) Epidemiologic approach to the etiology of type 1 diabetes mellitus and its complications. N Engl J Med 317:1390–1398
Li JH, Wang W, Huang XR, Oldfield M, Schmidt AM, Cooper ME, Lan HY (2004) Advanced glycation end products induce tubular epithelial-myofibroblast transition through the RAGE-ERK1/2 MAP kinase signaling pathway. Am J Pathol 164:1389–1397
Li Y, Yang J, Dai C, Wu C, Liu Y (2003) Role for integrin-linked kinase in mediating tubular epithelial to mesenchymal transition and renal interstitial fibrogenesis. J Clin Invest 112:503–516
Liu Y (2004) Epithelial mesenchymal transition in renal fibrogenesis: pathologic significance, molecular mechanism, and therapeutic intervention. J Am Soc Nephrol 15:1–12
Lyamichev V, Mast AL, Hall JG, Prudent JR, Kaiser MW, Takova T, Kwiatkowski RW, Sander TJ, de Arruda M, Arco DA, Neri BP, Brow MA (1999) Polymorphism identification and quantitative detection of genomic DNA by invasive cleavage of oligonucleotide probes. Nat Biotechnol 17:292–296
Mein CA, Barratt BJ, Dunn MG, Siegmund T, Smith AN, Esposito L, Nutland S, Stevens HE, Wilson AJ, Phillips MS, Jarvis N, Law S, de Arruda M, Todd JA (2000) Evaluation of single nucleotide polymorphism typing with invader on PCR amplicons and its automation. Genome Res 10:330–343
Moczulski DK, Rogus JJ, Antonellis A, Warram JH, Krolewski AS (1998) Major susceptibility locus for nephropathy in Type 1 diabetes on chromosome 3q: results of novel discordant sib-pair analysis. Diabetes 47:1164–1169
Nakai S, Shinzato T, Sanaka T, Kikuchi K, Kitaoka T, Shinoda T (2002) The current state of chronic dialysis treatment in Japan. J Jpn Soc Dial Ther 35:1155–1184
Nielsen DM, Ehm MG, Weir BS (1998) Detecting marker-disease association by testing for Hardy–Weinberg disequilibrium at a marker locus. Am J Hum Genet 63:1531–1540
Ohnishi Y, Tanaka T, Ozaki K, Yamada R, Suzuki H, Nakamura Y (2001) A high-throughput SNP typing system for genome-wide association studies. J Hum Genet 46:471–477
Okada H, Danoff TM, Kalluri R, Neilson EG (1997) Early role of Fsp1 in epithelial–mesenchymal transformation. Am J Physiol 273:F563–F574
Oldfield MD, Bach LA, Forbes JM, Nikolic-Paterson D, McRobert A, Thallas V, Atkins RC, Osicka T, Jerums G, Cooper ME (2001) Advanced glycation end products cause epithelial-myofibroblast transdifferentiation via the receptor for advanced glycation end products (RAGE). J Clin Invest 108:1853–1863
Palczewski K, Polans AS, Baehr W, Ames JB (2000) Ca2+-binding proteins in the retina: structure, function, and the etiology of human visual disease. Bioessays 22:337–350
Parving HH, Tarnow L, Rossing P (1996) Genetics of diabetic nephropathy. J Am Soc Nephrol 7:2509–2517
Polans A, Baehr W, Palczewski K (1996) Turned on by Ca2+. The physiology and pathology of Ca(2+)-binding proteins in the retina. Trends Neurosci 19:547–554
Quinn M, Angelico MC, Warram JH, Krolewski AS (1996) Familial factors determine the development of diabetic nephropathy in patients with IDDM. Diabetologia 39:940–945
Risch N, Merikangas K (1996) The future of genetic studies of complex human disease. Science 273:1516–1517
Shimazaki A, Kawamura Y, Kanazawa A, Sekine A, Saito S, Tsunoda T, Koya D, Babazono T, Tanaka Y, Matsuda M, Kawai K, Iiizumi T, Imanishi M, Shinosaki T, Yanagimoto T, Ikeda M, Omachi S, Kashiwagi A, Kaku K, Iwamoto Y, Kawamori R, Kikkawa R, Nakajima M, Nakamura Y, Maeda S (2005) Genetic variations in the gene encoding ELMO1 are associated with susceptibility to diabetic nephropathy. Diabetes 54:1171–1178
Susztak K, Sharma K, Schiffer M, McCue P, Ciccone E, Bottinger EP (2003) Genomic strategies for diabetic nephropathy. J Am Soc Nephrol 14:S271–S278
Suzuki A, Yamada R, Chang X, Tokuhiro S, Sawada T, Suzuki M, Nagasaki M, Nakayama-Hamada M, Kawaida R, Ono M, Ohtsuki M, Furukawa H, Yoshino S, Yukioka M, Tohma S, Matsubara T, Wakitani S, Teshima R, Nishioka Y, Sekine A, Iida A, Takahashi A, Tsunoda T, Nakamura Y, Yamamoto K (2003) Functional haplotypes of PADI4, encoding citrullinating enzyme peptidylarginine deiminase 4, are associated with rheumatoid arthritis. Nat Genet 34:395–402
Tanaka N, Babazono T, Saito S, Sekine A, Tsunoda T, Haneda M, Tanaka Y, Fujioka T, Kaku K, Kawamori R, Kikkawa R, Iwamoto Y, Nakamura Y, Maeda S (2003) Association of solute carrier family 12 (sodium/chloride) member 3 with diabetic nephropathy, identified by genome-wide analysis of single nucleotide polymorphisms. Diabetes 52:2848–2853
Terasawa M, Nakano A, Kobayashi R, Hidaka H (1992) Neurocalcin. A novel calcium-binding protein from bovine brain. J Biol Chem 267:19596–19599
The International HapMap Consortium (2005) A haplotype map of the human genome. Nature 437:1299–1320
Vardarli I, Baier LJ, Hanson RL, Akkoyun I, Fischer C, Rohmeiss P, Basci A, Bartram CR, Van Der Woude FJ, Janssen B (2002) Gene for susceptibility to diabetic nephropathy in type 2 diabetes maps to 18q22.3-23. Kidney Int 62:2176–2183
Vijay-Kumar S, Kumar VD (1999) Crystal structure of recombinant bovine neurocalcin. Nat Struct Biol 6:80–88
Wang W, Zhou Z, Zhao W, Huang Y, Tang R, Ying K, Xie Y, Mao Y (2001) Molecular cloning, mapping and characterization of the human neurocalcin delta gene (NCALD). Biochem Biophys Acta 1518:162–167
Zozulya S, Stryer L (1992) Calcium-myristoyl protein switch. Proc Natl Acad Sci USA 89:11569–11573
Acknowledgments
We thank A. Kanazawa, S. Tsukada and the technical staff of the Laboratory for Diabetic Nephropathy at the SNP Research Center. This work was partly supported by a grant from the Japanese Millennium Project. Support for Ng DPK was provided by the National Medical Research Council (Singapore) (NMRC/1018/2005). All authors have no conflict of interest, and nothing to declare.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kamiyama, M., Kobayashi, M., Araki, Si. et al. Polymorphisms in the 3′ UTR in the neurocalcin δ gene affect mRNA stability, and confer susceptibility to diabetic nephropathy. Hum Genet 122, 397–407 (2007). https://doi.org/10.1007/s00439-007-0414-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-007-0414-3